To download or print this page in a different language, choose your language from the drop down menu in the upper left of the website first. If you would like the references to print along with the position paper, please click the “References” area at the bottom of the article to expand them into view and then click the print button.

State of the Science on Root Canal Treated Teeth (RCTT)
Compiled, Developed, Written, and Released by
Jack Kall, DMD, FAGD, MIAOMT
Teri Franklin, PhD, Chief Science Writer, IAOMT
The IAOMT wishes to recognize the valuable contributions of
Dr. Valerie Kanter, DMD MS BCN
Released:
Approved by the IAOMT Science Committee: March 12, 2026
Approved by the IAOMT Board of Directors: March 12, 2026
Disclaimer: The IAOMT has used scientific evidence, expert opinion, and its professional judgment in assessing this information and formulating this paper. No other warranty or representation expressed or implied, as to the interpretation, analysis, and/or efficacy of the information is intended in this paper. The views expressed in this document do not necessarily reflect the views of the IAOMT’s Executive Council, Scientific Advisory Board, administration, membership, employees, contractors, etc. This report is based solely on the information the IAOMT has obtained to date, and updates should be expected. Furthermore, as with all guidelines, the potential for exceptions to the recommendations based upon individual findings and health history must likewise be recognized. IAOMT disclaims any liability or responsibility to any person or party for any loss, damage, expense, fine, or penalty which may arise or result from the use of any information or recommendations contained in this report. Any use which a third party makes of this report, or any reliance on or decisions made based on it, are the sole responsibility of the third party.
Table of Contents
- Summary
- What Is Root Canal Treatment (RCT)?
Figure 1. Root Canal Treatment - Apical Periodontitis (AP)
- Is Root Canal Treatment Safe and Effective?
Defining ‘Safe’ and ‘Effective’
Limitations in the Literature regarding the prevalence of CAP
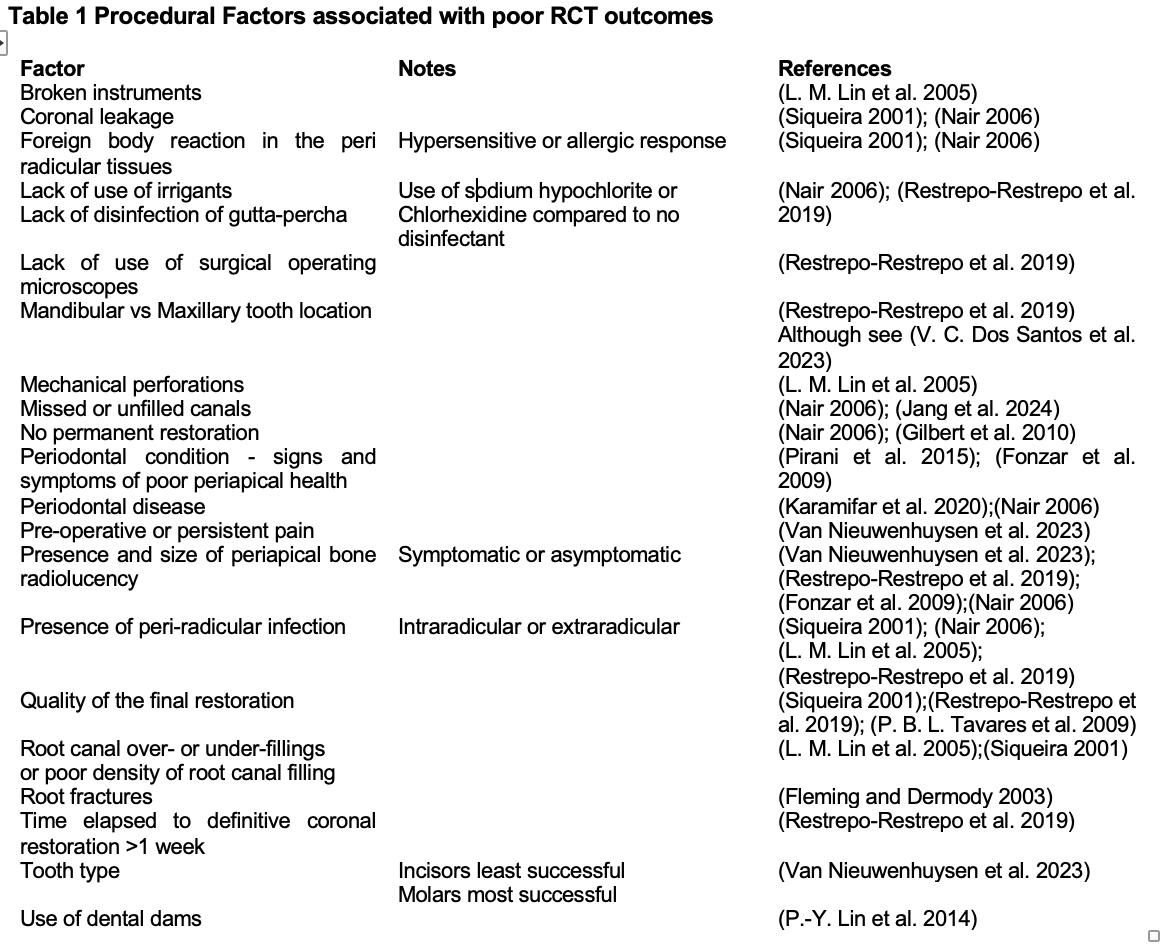
Table 1. Procedural Factors associated with poor RCT outcomes
Patient Risk Factors
Figure 2. Root Anatomy
Potential Health Benefits of Successful RCT - RCT and Associations with Systemic Disease
Table 2. Associations between AP and Systemic Disease - RCT: Animal and Preclinical Studies
- Special Populations
- Treatment Alternatives to Root Canal Therapy
- New and Emerging Technologies in Endodontics
Cone Beam Computed Tomography (CBCT)
Ozone Therapy
Activated Irrigation in Endodontics
Figure 3. Images of root penetration of irrigants using different methods
Figure 4. Graph of root penetration of irrigants using different methods
Photobiomodulation (PBM)
Platelet-rich fibrin (PRF)
Potential Future Strategies
* Nutritional and Lifestyle Interventions
* Next-generation antibacterial strategies (NGAS) - Deciding Whether to Retain or Remove Root Canal–Treated Teeth
- Conclusions
- References
1. Summary
This IAOMT paper reviews the current state of the science on root canal treated teeth with a focus on chronic apical periodontitis (CAP), treatment limitations and outcomes, and potential systemic health implications. While root canal treatment (RCT) is widely used to manage infected or necrotic dental pulp, a substantial body of evidence demonstrates that post-treatment apical periodontitis remains highly prevalent, affecting approximately 40 – 60% of endodontically treated teeth. Advances in imaging, particularly cone beam computed tomography (CBCT), reveal that traditional two-dimensional (2-D) radiography significantly underestimates persistent periapical disease. Procedural factors, patient-specific risk factors, and anatomical complexities all contribute to incomplete disinfection and microbial persistence, calling into question commonly reported success rates for RCT.
The paper further synthesizes extensive epidemiologic, mechanistic, and animal research demonstrating strong associations between RCT-related CAP and a wide range of systemic conditions, including cardiovascular disease, diabetes, neuroinflammation, metabolic syndrome, autoimmune disorders, and adverse pregnancy outcomes. Proposed biological mechanisms include bacteremia, endotoxin-mediated inflammation, immune dysregulation, and oral/gut systemic interactions. While causation in humans cannot always be definitively established, animal studies consistently show deleterious systemic effects of CAP, strengthening the biological plausibility of these associations. This paper also discusses clinical decision-making regarding retention versus extraction of root canal treated teeth, special at-risk populations, emerging diagnostic tools, and newer technologies aimed at improving disinfection and healing outcomes.
2. What is Root Canal Treatment (RCT)?
Endodontic therapy, also known as endodontic treatment or root canal therapy (RCT), is a treatment sequence that removes inflamed, infected and/or necrotic pulp (frequently called the nerve) from within the root(s) of a tooth, combined with disinfection and the protection of the decontaminated tooth from future microbial invasion. This is accomplished by a cleaning process associated with disinfection that includes chemical, and sometimes ultrasonic and/or laser techniques that are part of the irrigation process. Mechanical enlargement of the main canal diameter enhances the elimination of the smear layer, and provides a better access to the dentin walls, accessory canals and dentinal tubules. The final step is the obturation or filling process usually with gutta percha and a root canal sealer.1,2
Figure 1
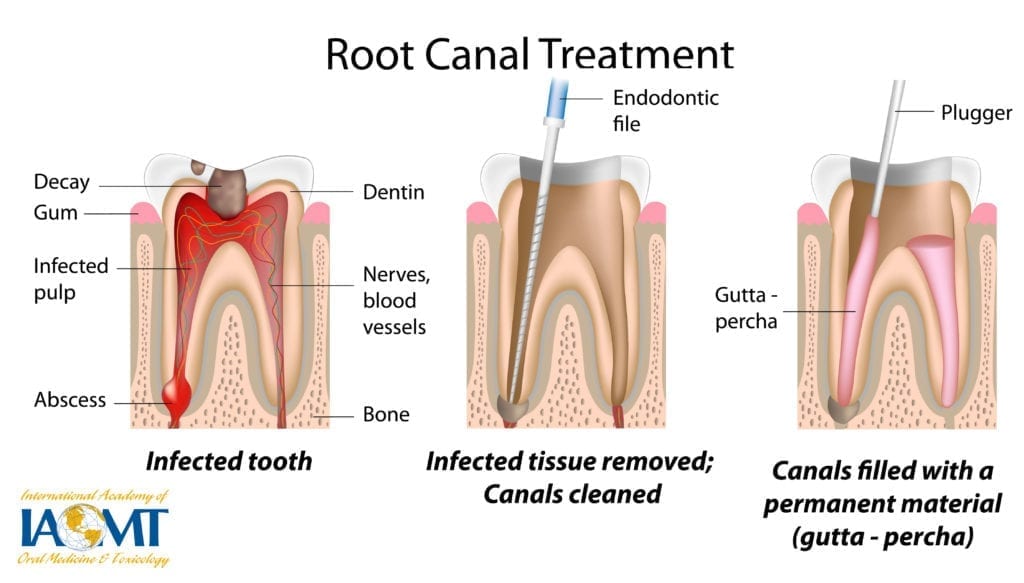
Image courtesy of Jack Kall, DMD
Typically, a dental specialist known as an endodontist carries out RCTs however in some cases, general dentists also perform them. Endodontics is the branch of dentistry that focuses on the treatment of the dental pulp, the soft tissues inside the tooth (nerves, blood vessels and connective tissue). The goal of endodontic therapy, or RCT, is to prevent and/or treat infections of these structures. The diagnosis of AP is often made by observing a periapical radiolucency, the radiographic sign of inflammatory bone lesions around the apex of the tooth root, which is an indicator that RCT or extraction may be warranted.
3. Apical Periodontitis (AP)
As stated above, AP is an acute or chronic inflammatory lesion around the end of the root apex. It is characterized by inflammation, destruction, and/or resorption of bone located around the dental apex. Although it is a local infection, pathogens and their products in the periapical area, as well as inflammatory cytokines, reach other regions of the body and can trigger a systemic immune/inflammatory response in the host.3 This condition has been associated with several types of systemic diseases.4
AP is typically the diagnosis that leads to either tooth extraction or RCT if the patient wants to keep a structurally sound and restorable (while nonvital) tooth. Even in instances wherein all precautions were observed and excellent procedural RCT was conducted, AP can persist and may not completely resolve even when previous symptoms, such as pain, or infection no longer persist.
Citing older reports, the American Association of Endodontics (AAE), state on their website that the prevalence of AP in teeth that have previously undergone RCT ranges from 40%–61%.5 A recent review corroborates these figures and suggests that the numbers are rising, and are greater in individuals with inadequate restorative and endodontic treatment: AP was found in 41% of adults with RCT-treated teeth compared to 2% in untreated teeth, suggesting that prior RCT underlies the diagnosis of AP. In addition, sex-specific effects were found wherein AP in previously treated teeth was more prevalent in men.6 And yet, the AAE state on their website: “The AAE holds that there is no valid scientific evidence linking endodontically treated teeth to systemic diseases. The AAE maintains that RCT is safe, effective, and, along with proper restoration, helps patients retain natural teeth. The theory connecting root canals to disease is based on long-discredited research.” It is clear that the AAE’s position on the safety and effectiveness of RCT is inadequate to address potential systemic complications that can occur.
If the global prevalence of people with at least one root canal treated tooth is 56%7 and if half of those people have RCT-related AP,6 this translates to 1 in 4 adults with RCT-related AP. These are staggering numbers pointing to AP as existing in epidemic proportions. Biological dentistry examines all of the available scientific evidence to formulate guidelines and conclusions on this crucial and important health problem of AP in endodontically treated teeth (for more on biological dentistry see An Introduction to Biological Dentistry).
In the early 1900s, Canadian dentist Weston A. Price proposed the “focal infection theory,” which suggested that bacteria lingering in the roots after RCT could migrate to other areas of the body and lead to systemic illness, including cancer, arthritis, cardiovascular, and kidney disease. Dr. Price conducted hundreds of experiments confirming his hypothesis. Price’s experiments, while somewhat primitive, were published in peer-reviewed journals, indicating that they underwent rigorous scientific scrutiny. In 1951, the American Dental Association (ADA) published a review of Price’s methods in their dental journal, Journal of the American Dental Association (JADA), stating that Price’s methods lacked the rigor of modern science, including proper control groups. However, the publication is not a review, and reads more like an editorial, i.e., an opinion.8,9
In 2019, Netflix released a documentary entitled Root Cause, that led to concern among the general public and created unrest among many dental professionals. The film chronicles the 10-year search of a young man trying to regain his health and finding in the end that his RCTT were making him sick. Many in the dental community condemned the film as inaccurate and labeled it as disseminating malicious misinformation.10 Nonetheless the documentary spiked new debate over RCT which has reached a critical juncture. Concerns about this dental procedure and its potential impact on the rest of the body are being raised by patients and providers, as well as dental schools and organizations. One position is to condemn all RCT disregarding the evidence of its utility in reducing infection and providing symptom relief, while the opposing position is to declare RCT as safe without concern for potential lingering infection and long-term systemic effects. The goal of this IAOMT paper is to review the state of the science at this juncture, in the year 2026. A key word literature search was conducted using PUBMED (Medline is included within PUBMED). PUBMED provides articles, in order, based on ‘best match’ with key words. Click to view the search strategy, which spanned 2000-2025 and for the reference list, which includes 561 references.
4. Is RCT safe and effective?
Defining ‘Safe’ and ‘Effective’
The question arises; do we agree with the ADA and AAE position that RCT is safe and effective? The simple answer is no – but this is more likely related to the definitions used by dental associations versus biological dentistry’s definitions of ‘safe’ and ‘effective’.
Generally, safety refers to 1) risk of serious adverse events; 2) risk of contributing to systemic disease; and 3) risk compared to alternative treatments. In the eyes of the AAE, if 1) no adverse events are noted (i.e., patient does not have pain); 2) a consensus statement on the procedure’s contributing effects on systemic disease is lacking; and 3) the risk appears lower than alternative treatments (i.e., tooth extraction, which is a decision specific to each case); then the AAE may use the word ‘safe’ to describe RCT. As biological dentists we may view the procedure as unsafe because 1) there is a high chance of persistent AP (i.e., underlying infection) regardless of whether the patient experiences pain; 2) the hundreds of human studies showing associations with systemic disease and hundreds of animal studies showing biological plausibility; and 3) the probability that tooth extraction, which removes all of the infected tooth does not lead to CAP.
Large organizations upon which people generally turn to for information and guidance use core principles to determine the effectiveness of a technique. Thus, if properly diagnosed, performed, restored and followed up, RCT healing rates appear to be high and therefore efficacy appears to be high, which may be translated by the organization into a measure of effectiveness. But in real-world conditions, across general populations, and including variability in the talent/expertise of the specialist performing the procedure and multiple other variables described in Table 1, CAP rates are much greater,5 which may imply less effectiveness from a biological dentistry perspective.
Bottom line – in cases of persistent AP following RCT, biological dentists might consider the treatment unsafe and ineffective, while if the lesion is smaller and the patient is not experiencing pain, the AAE may consider the procedure safe and effective. Further, it is becoming more widely recognized, that if the health of the mouth is compromised, so is the health of the body. The mouth is the gateway to the digestive tract – what happens in the oral cavity can impact our entire body. Please visit the IAOMT’s Oral Health Integration and Biological Dentistry to learn more about the mouth/body connection.
Limitations in the Literature regarding the prevalence of CAP
Even though the literature shows that the prevalence of AP in endodontically treated teeth is at least 40%,5 6 Wu et al suggest that there are multiple limitations of previously published studies and systematic reviews evaluating the outcome of RCT. Traditionally, 2-D periapical radiography has been used to assess RCT outcomes, with the absence of a periapical radiolucency implying a healthy periapical area. In a review article 57% of studies used both clinical and radiographic findings to determine treatment outcome. As post-treatment AP is often asymptomatic, the outcome was determined by radiographic examination alone in the remaining 43% of studies. Either strict (complete resolution of existing periapical radiolucency at recall) or loose (reduction in size of existing periapical radiolucency at recall) 2-D radiographic criteria were used in these studies.11 However, a high percentage of cases confirmed as healthy by 2-D radiographs revealed AP with cone beam computed tomography (CBCT) imaging and by histology. In teeth where reduced size of the existing radiolucency was diagnosed by 2-D radiographs and considered to represent periapical healing, CBCT showed enlargement of the lesion.11
In clinical studies, two additional factors may have further contributed to the overestimation of successful outcomes after RCT: 1) extractions and re-treatments were rarely recorded as failures; and 2) post-treatment patient contact was often lower than 50%, both indicators that RCT was not successful. Further, the periapical index, frequently used for determination of success, was based on radiographic and histological findings in the periapical region of maxillary incisors. The validity of using this index for all tooth positions is questionable, as the thickness of the cortical bone and the position of the root tip in relation to the cortex varies depending on tooth position. Therefore, systematic reviews reporting RCT success rates without referring to these limitations are misleading. Long-term longitudinal studies using CBCT and stricter evaluation criteria are necessary to correctly evaluate RCT outcomes.12
Some of the factors associated with poor outcomes of RCT that are associated with the procedure itself are listed in the table below. The quality of the treatment and final restoration are strong predictors.
Patient risk factors Figure 2 Root Anatomy
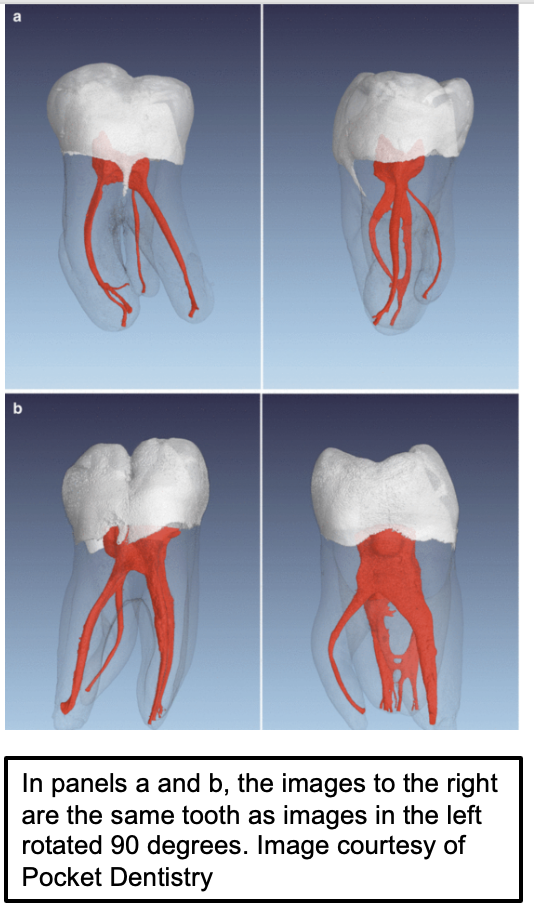
Patient risk factors also impact outcomes. At the patient level, risk factors include but are not limited to chronic alcohol use,27 cigarette smoking,28,29 high carbohydrate diet,30 pre-existing diabetes,31 medication use,32 and metabolic syndrome.33 One of the biggest predictors is advancing age.17,29 but one must consider that advancing age is strongly associated with more health issues and medication use. Evidence also exists for genetic control over responses to bacterial accumulation which could lead to AP, but more study is needed in this area.34 The complexity of root canal systems further complicates treatment outcomes. It has been shown that anatomical variations, such as accessory canals, can harbor necrotic pulp tissue accompanied by even more bacteria that may contribute to persistent infections if not properly addressed during therapy.35 Figure 2 provides an example of the complexity of root canal systems).
Potential Health Benefits of Successful RCT
Although much less prevalent, evidence exists supporting the systemic benefits of successful endodontic treatment. One study of 15 individuals with AP found that RCT reduced levels of the inflammatory biomarker, high-sensitivity C-reactive protein (hs-CRP), and showed that 10 of the 15 patients reduced their CVD risk status following RCT.36 Of note, 6 months after RCT, the average periapical index score (PAI) had significantly reduced from approximately 3.2 to 1.4. Another much more rigorous study conducted by Kumar et al (2022) is supportive. hs-CRP levels were compared before and after RCT in otherwise healthy patients with asymptomatic AP (n=25) and matched healthy controls (n=25). hs-CRP was significantly higher in the AP group at baseline. In the AP group, at 6-months follow-up (n=22) hs-CRP was significantly reduced, suggesting reduced CVD risk. PAI scores were significantly reduced from approximately 3.9 to 2.3.37
There are further studies that ‘state’ that successful RCT leads to reductions in inflammatory biomarkers, but closer examination reveals ambiguity. For example, a series of studies began by first observing elevated inflammatory biomarkers of CVD risk in people with AP compared to healthy people.38 Next, the authors found that the inflammatory biomarkers in the microbiome of saliva, blood and intracanal samples of AP individuals were correlated, suggesting that AP infection could disseminate from the root canal to the body through the blood, potentially contributing to the inflammatory burden.39 RCT was performed on these same AP subjects. At 2 years, over half the subjects (37) came back for follow-up and of those, the authors claim 100% success, confirmed by clinical and 2-D radiographic reports: 21 cases were completely healed, and 16 cases were healing. Four CVD risk biomarkers were significantly reduced, while nine others had increased and a few were unchanged.40
So, while successful endodontic treatment reduced serum levels of some CVD risk biomarkers, hs-CRP, asymmetric dimethylarginine, and matrix metalloprotease-2, many others including IL-1β, IL-6 and MMP-8 were increased.40 The authors titled this work ‘Successful endodontic treatment reduces serum levels of cardiovascular disease risk biomarkers –high-sensitivity C-reactive protein [as above, hs-CRP], asymmetric dimethylarginine, and matrix metalloprotease-2’, without mention that over twice as many CDV risk factors were increased.
Another study examined 44 metabolites at baseline at 4 timepoints (3 months, 6 months, 1 yr and 2 yr). One hypothesis tested was that RCT would improve glucose levels. At the 2-year follow-up, glucose levels were reduced. A second tested hypothesis was that RCT would improve lipid metabolism. After RCT, cholesterol levels were significantly lower at the 3- and 6- month follow-ups when compared to baseline level. Choline levels were reduced at the 6-month follow-up. Fatty acid levels were reduced at the 3-month follow-up but increased at the 1- and 2-year follow-ups. Triglyceride levels showed no significant changes at the 3- and 6-month follow-ups but increased at the 1- and 2-year follow-ups. The authors conclude that “together, these findings highlighted a link between successful endodontic treatment and a short-term benefit on lipid metabolism”.41
Perhaps that conclusion is partially true, however, it is well-accepted in the medical field that the most sensitive and clinically meaningful indicator of a poor lipid metabolism profile is high triglycerides.42–45 Elevated triglycerides are a core marker of dyslipidemia and metabolic dysfunction. They reflect insulin resistance and impaired fatty acid oxidation and are strongly associated with metabolic syndrome and CVD risk.42–45 It is also well-known that measuring cholesterol, without distinguishing between high and low density lipids is insufficient. Normal cholesterol levels can be observed in someone with severe metabolic dysfunction.46,47 While the observation of improved glucose levels is positive, the lack of discussion regarding the increase in triglycerides is concerning.
A third study aimed to determine whether RCT led to reductions in biomarkers. This paper is entitled “Reduced C-reactive protein levels after root canal treatment in clinically healthy young apical periodontitis individuals at cardiovascular risk. A prospective study.” This study examined multiple biomarkers at 1- and 6-months post-RCT in 29 individuals. In all biomarkers, except one, no significant differences could be observed. hs-CRP was reduced at 1 month but rose again by 6 months. Half of the individuals (15) were identified at baseline as having CVD risk. Of those, only 7 returned for the 6-month evaluation. Within these individuals hs-CRP was reduced at both time points. PAI scores were reduced from approximately 5 at the first follow-up visit to 3 but not reduced further at 6-months. There are several limitations of this study, including not controlling for multiple comparisons (i.e., false positives due to the number of variables that were assessed), and the very small number of subjects who returned for the 2nd evaluation within the CVD risk group.48
In some of the studies outlined above, PAI scores were reduced, showing some clinical benefit. Nonetheless, while the authors of this RCTT paper attempted to elucidate the potential systemic health benefits of RCT, what was found was that claims of health benefits remain unsubstantiated.
At times the choice is made to have an endodontic procedure. Perhaps the alternatives, described in detail below and that usually begin with tooth extraction, are not an option for the patient due to medical, aesthetic or financial reasons. Sometimes the patient is just not emotionally ready to lose a tooth, even a nonvital one. If important considerations are taken into account, RCT may be a workable option for some people.
5. RCT and Associations with Systemic Disease
Hundreds of studies show associations between AP and systemic health issues, including CVD, diabetes and others. Keeping in mind that prospective longitudinal controlled trials are necessary to show causation, rather than association. Such studies are nearly impossible to conduct in humans. Thus, scientific study depends on well-conducted association studies and preclinical (animal) studies.
For example, in over 2 million patients significant associations were found between the presence of endodontic pathology and a history of hypertension, myocardial infarction, cerebrovascular accident, congestive heart failure, heart block, deep vein thrombosis, and cardiac surgery.49 Table 2 presents review articles on this subject that cite the original research for the interested reader. Important to consider when evaluating these studies is the hypothesis being tested – were the authors asking: Is AP associated with systemic disease?’ or ‘Is systemic disease associated with AP?’ It is also important to keep in mind that there are likely two sides to the coin: While systemic disease may influence the pathogenesis of endodontic infection, endodontic infection can also influence systemic health. For example, a systematic review indicated that chronic diseases, including diabetes and hypertension, can impair healing outcomes following endodontic treatment, leading to a higher prevalence of persistent infections and complications post-RCT.50 While other studies indicate that the presence of AP may exacerbate systemic conditions such as type 2 diabetes, potentially impairing wound healing after endodontic treatment.33 Because of this likely bidirectional relationship, Endodontic Medicine has gained prominence in the field of endodontics.51
The mechanisms underlying the association between AP and systemic disease may be mediated, at least in part, by endotoxin.52 Endotoxin is a type of lipopolysaccharide, a major component of the outer membrane of gram-negative bacteria, which have a high degree of resistance to antibiotics and therefore can be extremely dangerous to human health. This outer membrane expresses a potent immune response inducer.53 Endotoxin was named such because it was originally thought to be a toxin confined within the bacteria. However, we now know that endotoxin is released from bacteria. Its toxicity is due to the host’s inflammatory reaction to it rather than an intrinsic toxicity to the bacteria itself.54 All organisms have some level of endotoxin within their bodies. In mammals, high levels are found in the gut. They are also found in saliva, dental plaque, skin, lungs, respiratory and urinary tracts and the blood. When they are found in blood, even at nanogram levels, it is usually a sign of infection. As described in Gomes et al (2018), endotoxin induces inflammatory responses through a complex pathway, activating hundreds of inflammatory genes. However, in the absence of infection, endotoxin is still able to cross mucosal membranes of the gut, gums, nose and/or the lungs. Gomes and colleagues showed that increased root canal endotoxin levels are associated with AP, oxidative and nitrosative stress, and the brain disease and severity of major depression.54 Endotoxin has been associated with neurodegenerative diseases such as Parkinson’s and Alzheimer’s.52,55,56 People with chronic gum disease have elevated blood endotoxin57 and a higher risk of Alzheimer’s disease and a faster rate of cognitive decline.58 It is not yet fully known how endotoxins enter the brain and lead to neurodegeneration but study is ongoing.52

6. RCT: Animal and Preclinical Studies
A review of Table 2 (associations between AP and Systemic Disease) is eye-opening but doesn’t give us information about causation. However, AP is associated with a complex bacterial microflora consisting of approximately 200 species.76 The anatomic closeness of this microflora to the bloodstream can facilitate bacteremia, the spread of bacteria and its components to the blood. Indeed, bacteremia, was observed in 18 to 54% of patients following RCT.77 This is further evidence that AP can cause disease but still is not definitive.
Three mechanisms or pathways linking oral infections, such as AP to secondary systemic effects have been proposed: 1) bacterial metastasis to the body from the oral cavity, 2) metastatic injury from the effects of circulating oral microbial toxins, and 3) metastatic inflammation caused by immunological injury induced by oral microorganisms.78 Although less is known about which pathway is taken, well-controlled animal studies, wherein AP is induced in animals can provide important supplementary information that should be taken into consideration when examining the science. For example, to examine whether AP could cause atherosclerosis, AP was induced in two groups of genetically modified (apolipoprotein E-deficient) mice: One group was fed a normal diet and the second group was fed a high fat diet to induce atherosclerosis. After 4 months the mice were euthanized. Both groups developed atherosclerosis regardless of diet,79 which provides useful information but is also limited in that it did not include a control group that did not have AP.
Thus, Gan et al conducted a similar experiment wherein AP was induced in one group of mice and compared to a control group without AP. A significant increase in atherosclerotic plaque formation was observed in the aortic arches of the AP mice compared to the control group.80 A secondary finding was that AP altered gut microbiota. Gan and colleagues continued this line of work showing significant changes of the gut microbiota: harmful bacterial species thrived while beneficial species declined. Disruptions were observed in lipid metabolism and bile acid synthesis, leading to elevated levels of harmful acids, potentially contributing to the development of atherosclerosis. Increased intestinal permeability was positively correlated with the severity of atherosclerotic lesions, highlighting the importance of the intestinal barrier in cardiovascular health. Not only does this preclinical line of research support the position that AP can cause coronary heart disease, but it underscores the intricate interplay between oral health, gut microbiota composition, and the incidence of cardiovascular disease.81
Other animal work examined how AP affects inflammation and early aortic lesions in rats fed a high fat diet. Rats were divided into 4 groups to examine the interacting effects of obesity and AP. Compared to rats without AP or high fat diet, AP increased several inflammatory markers, including serum IL-2, IL-6, and IL-10, while high-fat diet elevated MCP-1, TLR-4, and NF-κB p65 expression. Rats with both high fat diet and AP showed further upregulation of TLR-4. Findings suggest AP promotes systemic inflammation and may contribute to early aortic injury, highlighting its potential role in cardiovascular disease.82 Mechanistic studies are supportive of these findings, and point to the activation of more serious aortic inflammatory cytokine micro RNA expression.83
Considering that 12-15% of the American population have diabetes, and reiterating that 1 in 4 adults have AP, research is being conducted to examine the role of AP in diabetes. Interleukin-17 (IL-17) is a potent proinflammatory cytokine that is elevated in both Type 1 and Type 2 diabetes and contributes to insulin resistance, beta cell dysfunction, and diabetic complications. Higher IL-17 levels are associated with the presence and severity of diabetes and its related complications. Cintra and colleagues specifically examined serum IL-17 levels of normoglycemic and diabetic rats with and without AP. Regardless of diabetic status, AP significantly increased IL-17 serum levels. Negative outcomes in other measures were also observed in the rats with diabetes but without AP, which is not unexpected. These results support the position that AP leads to increases in inflammatory markers that signal diabetes.84 Other animal studies support the presence of higher inflammatory markers in AP.85 86
According to a systematic Global Burden of Disease study, approximately 1 in 6 adults are estimated to have neurological disease.87 This astronomical figure continues to grow. Perhaps the fact that approximately 1 in 4 adults now have AP related to RCT, may partially explain the increase in neurological disease. At least 3 different preclinical research groups have studied the effects of AP on the brain. Simões and colleagues showed an increase of the pro-inflammatory cytokines tumor necrosis factor-alpha (TNF-α), interleukin 1 beta (IL1β), and interleukin 6 (IL-6) in the hippocampus and frontal cortex of the brain.88 A second study observed brain hemorrhaging when AP was induced by specifically infusing Streptococcus mutans into the pulp tissue.89 And yet, another study focusing on perinatal effects, induced AP in pregnant rats and observed increases in inflammatory markers ( IL-6, TNF-α, and IL-1-b) in the brains of the pups,90 suggesting epigenetic influences.
Bain et al (2009) also examined the effects of AP on pregnancy outcomes. Compared to pregnant rats without AP, rats with AP had a significantly longer pregnancy and delivered pups with significantly higher birthweight. They also had significantly higher concentrations of IL-6, VEGF, IL-1-beta, and IL-10 within the uterine horn and IL-6, CRP and TNF-alpha within the liver (p<0.01). Blood glucose and serum TNF-alpha, IL-6, endothelin-1, IL-10, and insulin concentrations were significantly higher in the AP pregnant animals. The significant increase in serum TNF-alpha, blood glucose and serum insulin concentrations, suggest that AP animals developed insulin resistance, affecting their pregnancy outcomes.91
Other animal work has examined the impact of AP on rheumatoid arthritis (RA). Compared to animals with RA only, animals with both RA and AP had greater joint inflammation and severity (larger paw/knee circumference, more severe bone erosions and deformities); worse bone parameters, including reduced bone volume and density, fewer trabeculae, and greater trabecular separation; and an altered inflammatory profile, with higher TNF-α, lower IL-2, and elevated IL-17. The authors conclude that AP worsens RA progression and severity, suggesting a link between oral infection/inflammation and systemic autoimmune disease.92
While only a handful have been presented here, the preclinical (i.e., animal) field is exploding with new studies demonstrating the deleterious effects of AP on systemic health. These types of studies are unethical to conduct in humans and could not be ‘controlled’ to the same extent and yet, are crucial to our understanding, and point directly to causal mechanisms. To name a few, studies on impaired heart function in hypertensive rats with AP93; cardiac function in diabetic rats with AP94; potentiated consequences of diabetes in rats with AP95; inflammatory cytokines in the blood of AP rats96,97; impacts on metabolic disorders98; and AP-activated systemic and liver inflammation99. One animal study specifically evaluated the potential bidirectional influence of metabolic syndrome and AP and found that AP worsened metabolic syndrome, while metabolic syndrome had no effect on AP.100
In conclusion, this IAOMT scientific enquiry was unable to find an animal study that did not show negative effects of AP on health parameters.
7. Special Populations
The IAOMT does not hold the position that all non-vital teeth should be extracted. However, it is clear that non-vital teeth, with or without endodontic therapy, can present a systemic health risk in a significant portion of people. But who are these people and what percent is at risk? Throughout this paper we have identified several subgroups of people who are at risk to worsen their condition from RCT. For example, the 1 billion people (1/4 of the world’s population) who have metabolic syndrome101 are at risk. This is increased by the 12-15% of people with diabetes102 and the 48% who have hypertension.103 A thorough medical history conducted by the dentist will identify a significant number of U.S. adults as ‘at risk’. Dentists and endodontic specialists should approach treatment plans with careful consideration of the patient’s overall health status to optimize both dental and systemic health outcomes.104 Subsequently, each patient must be evaluated on an individual basis, considering their medical status and other factors.
8. Treatment Alternatives to RCT
When the pulp of a tooth becomes irreversibly damaged or infected (i.e., nonvital tooth), in most cases, the most definitive alternative to RCT is complete tooth extraction, which avoids the long-term risk of a RCTT continuing to harbor bacteria, and blocks the continued spread of harmful bacteria throughout the body. Once extracted, the missing tooth can be replaced with a dental implant, a fixed dental bridge or a removable partial denture. An implant is a durable, standalone solution where a root substitute (either titanium or ceramic) is surgically placed in the jawbone to support a crown. This prevents bone loss and preserves the alignment of surrounding teeth. The IAOMT is of the current opinion that ceramic zirconia implants, which are metal-free, are the most biocompatible alternative. A fixed dental bridge uses crowns on adjacent teeth to support an artificial tooth that “bridges” the gap. Removable partial dentures offer a non-surgical and cost-effective method for replacing one or more missing teeth. However, to preserve bone structure and health, and prevent the teeth from shifting, an implant consisting of a biocompatible material (ideally ceramic), covered by a zirconia crown is the preferred recommendation of the IAOMT.
For teeth with milder pulp inflammation (i.e., vital teeth), more conservative treatments, such as vital pulp therapy is an option.105 If the pulp is exposed due to trauma or deep decay, a medicated calcium-based material is placed directly over the pulp to stimulate healing and protect it. If decay has reached the pulp but the root is still developing, which may be the case in children, a pulpotomy may be performed to remove only the infected part of the pulp. Although more commonly used in pediatric dentistry, this procedure has shown promise in adults.106,107
9. New and Emerging Technologies in Endodontics
As is often the case, newer techniques and technologies frequently don’t have the track record or published studies confirming their possible benefits. Yet, due to the risk of less-than-ideal outcomes for RCT described in this report, individuals choosing to retain their tooth may want to consider seeking a dentist who utilizes some of the following adjuncts to traditional RCT, that may improve outcomes.
Cone Beam Computed Tomography (CBCT)
As referred to above, anatomical variations in the presence and number of canals in the root of a tooth are common (See Figure 2). The usual root canal anatomy of maxillary first molars consists of three roots with three canals, however up to six, and as few as two canals have been reported.108 Accessory canals are hard to visualize using 2-D radiography alone and can conceal necrotic pulp tissue contributing to persistent infections if not properly addressed.35,109 The use of technically advanced imaging modalities such as cone-beam computed tomography (CBCT) have been instrumental in pre-operative diagnosis to identify potential issues not visible with traditional 2-D X-rays, such as accessory canals.110 The cornerstone of CBCT technology is its ability to view a lesion of interest in 3-D (frontal, sagittal, coronal). The use of CBCT provides a higher degree of certainty (over 2-D radiographs) that all canals are treated, to avoid potential systemic health issues that could arise from traditional RCT. It is also useful in identifying early signs or spread of infection. Thus, the CBCT radiographic examination has become the gold standard for evaluating teeth and the surrounding bone.
Ozone Therapy
The utilization of advanced disinfection and healing techniques has improved outcomes for both RCT teeth and the alternatives.111,112 Ozone gas or ozonated water is a powerful disinfectant that kills bacteria, viruses, and fungi during dental procedures and aids in healing teeth, gums and bones. Staying within the accepted standard of care, and with proper application, oxygen/ozone can enhance the outcome in many aspects of dentistry.111 For example, in RCT re-treatment it can be used as a less traumatic procedure than tooth removal, to irrigate and clean the root canals; and it can also be applied to disinfect the surgical site after a tooth extraction. To learn more on the use of ozone therapy, click on IAOMT’s Ozone Therapy in Biological Dentistry.
Activated Irrigation in Endodontics
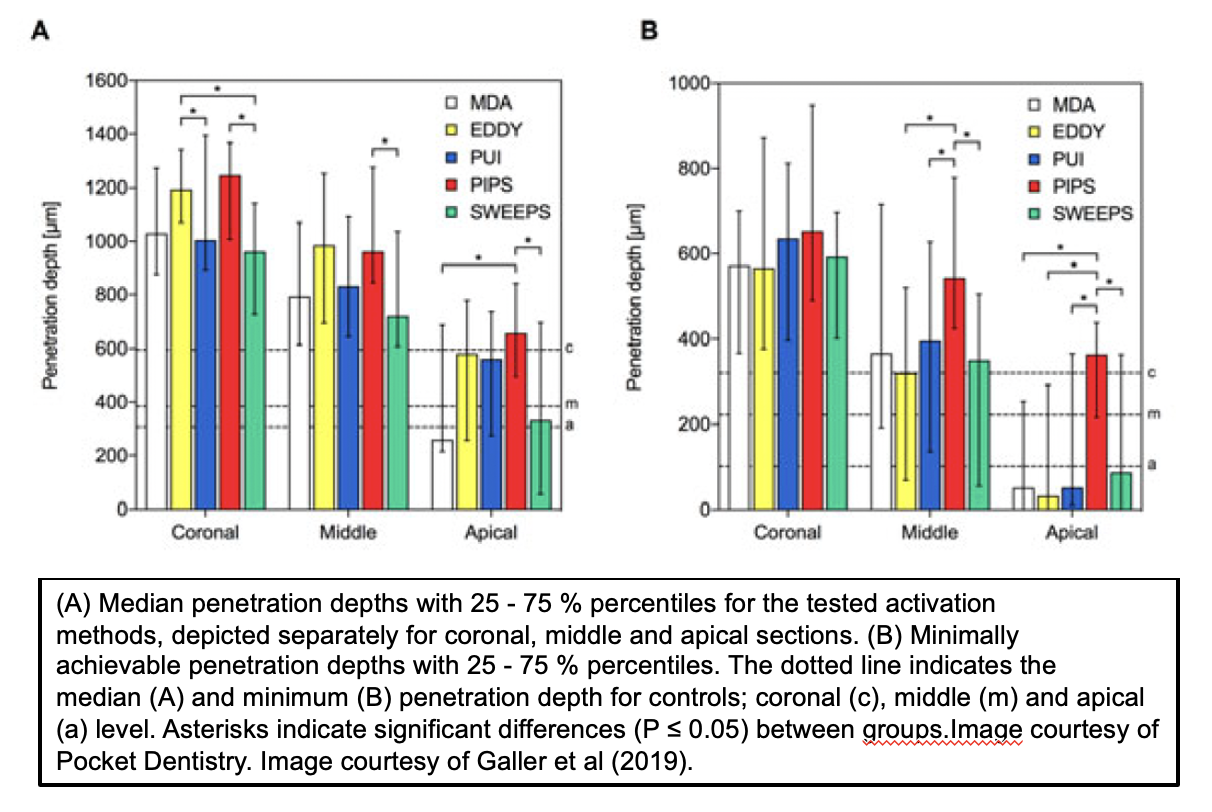
An important part of the cleaning process of the canals in the root is the flushing out of debris referred to as irrigation. As summarized by Mirza et al (2019), activated irrigation in endodontics has emerged in response to well-documented limitations of conventional syringe-and-needle irrigation.113 Classic and contemporary studies demonstrate that complex root canal anatomy – including fins, isthmuses, oval extensions, and apical irregularities, often remains inadequately cleaned by mechanical instrumentation alone, even with advanced nickel-titanium systems. Early work established that irrigant penetration and replacement are critical determinants of cleaning efficacy,114,115 while later micro-CT and morphologic studies confirmed that substantial portions of canal walls remain untouched after preparation.116–118 Because microbial persistence is a major determinant of endodontic failure, as shown in large outcome studies and meta-analyses (See Table 1), enhanced irrigant delivery and activation became a central focus. Chemical irrigants such as sodium hypochlorite and EDTA possess proven antimicrobial and tissue-dissolving properties, but their effectiveness is strongly influenced by fluid dynamics, renewal, and contact with biofilms, prompting the development of activated irrigation strategies.113
Activated irrigation techniques, including passive ultrasonic irrigation (PUI), sonic activation (GentleWave™), laser-activated irrigation (LAI), and photon-initiated photoacoustic streaming (PIPS and SWEEPS), significantly improve irrigant distribution, smear layer removal, and biofilm disruption through acoustic streaming and cavitation (See Figures 3 and 4).119 Ultrasonic activation has been shown to generate acoustic cavitation and enhanced debris removal, while erbium laser systems induce rapid vapor bubble formation and collapse, producing powerful fluid motion even in minimally enlarged canals. Visualization and experimental studies demonstrate superior penetration into lateral anatomy and apical regions compared with conventional irrigation. Numerous ex vivo and in vitro investigations report significantly greater reduction of Enterococcus faecalis biofilms when irrigants are activated ultrasonically or with lasers, particularly when combined with sodium hypochlorite. Importantly, laser-activated irrigation enables effective cleaning with lower irrigant volumes and reduced apical extrusion, aligning with the principles of minimally invasive endodontics. Collectively, the evidence supports activated irrigation as a critical adjunct to shaping, enhancing disinfection, improving safety, and potentially contributing to more predictable long-term endodontic success.113 Nonetheless, as can be observed in Figure 3, even using the best methods (i.e., PIPS) the irrigant has not completely penetrated the dentin, which would be optimal. Furthermore, penetration of the irrigant does not confirm the level of disinfection.
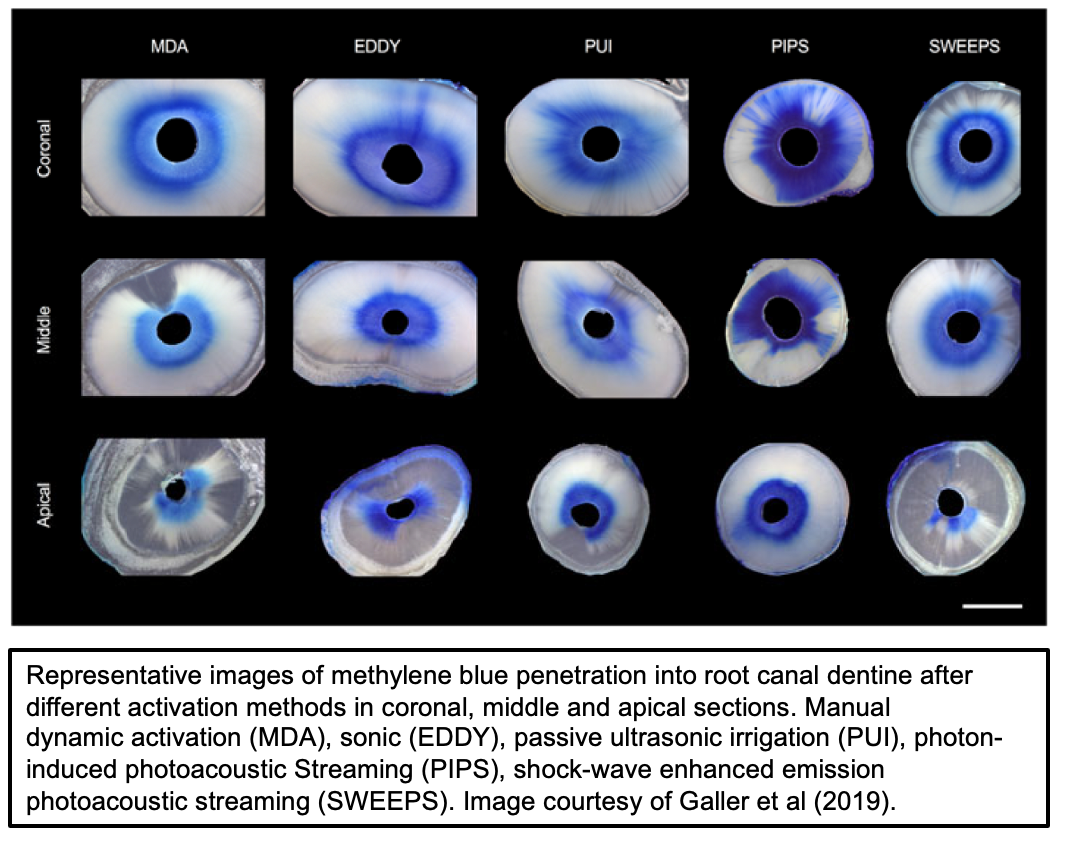 Figure 3 Images of root penetration of irrigants using different methods
Figure 3 Images of root penetration of irrigants using different methods
Figure 4 Graph of root penetration of irrigants using different methods
Photobiomodulation (PBM)
Another approach that can improve dental outcomes is the use of Photobiomodulation (PBM), which is also referred to as low-level laser therapy (LLLT).120 The radiation involved in generating laser light is nonionizing and therefore does not produce the same effects attributed to X-radiation. The FDA has approved the use of this technique to remove diseased gingival tissues and dental caries; as an aid when placing restorations; and as an adjunct treatment in root canal procedures, such as pulpotomies.121 PBM is a non-invasive, gentle and painless modality, and as such is a notable departure from some of the more aggressive treatment modalities commonly associated with dentistry.122
Platelet-rich fibrin (PRF)
PRF therapy is another adjunct treatment that has been shown to improve dental outcomes. This treatment involves a blood draw in the office, using a centrifuge to separate the blood into its individual components, thereby concentrating the patient’s own blood platelets to create a fibrin matrix, rich in growth factors. The PRF matrix consists of leukocytes, cytokines, and glycoproteins such as thrombospondin, and releases them for at least 7 days at the application site. Growth factors and released cytokines stimulate the activity of osteoblasts and accelerates tissue regeneration by increasing fibroblast migration.123 The use of PRF has shown to improve success rates in surgical cases such as sinus lift procedures, healing of extraction sockets, and management of AP.124 Indeed, a recent systematic review/meta-analysis on PRF in periapical surgery showed a positive association with reduced post-operative pain and improved radiographic healing,125 but more controlled trials are needed to confirm.126
Potential Future Strategies
Nutritional and Lifestyle Interventions
Other adjunct treatments are emerging and may prove promising at improving outcomes in the future. In one animal study both moderate physical activity and omega 3 supplementation improved CAP outcomes. Physical activity alone reduced inflammation through TNF-α modulation and controlling bacterial contamination. When combined with omega-3 supplementation, physical activity further improved inflammatory regulation by modulating IL-17 levels, reducing bone loss, and stimulating collagen production, thereby limiting inflammation and decreasing osteoclastic activity.127 Azuma and colleagues have also shown that omega-3 supplementation in AP rats decreases inflammatory mediators,128,129 and reduces bone resorption and promotes bone generation.130
Additional studies have examined the effects of supplementation on CAP. In one study, blood, saliva and root canals were examined in AP-induced rats supplemented with probiotics versus without. While no differences were observed between groups in blood and saliva profiles, within the canals inflammatory infiltrate and IL-1β and IL-6 in AP were significantly lower in the probiotic group compared with the control group.131 Oral supplementation with curcumin has also been shown to reduce AP severity in rats, suggesting an anti-inflammatory effect of curcumin on AP development.132 At least 4 animal studies have shown reduced inflammation and bone resorption in AP rats administered systemic melatonin compared to control animals.17,86,133,134 Melatonin has powerful antioxidant, anti-inflammatory, and bone-remodeling properties and may protect periodontal tissues by scavenging free radicals and modulating the immune response.135 Further interventional studies in humans are needed to confirm its effectiveness and establish appropriate application methods.
Next-generation antibacterial strategies (NGAS)
It cannot be repeated enough that despite standard root canal instrumentation and disinfection, failure rates are high due to persistent biofilms in complex canal anatomy. As described in a narrative review, next-generation antibacterial strategies (NGAS) using nanopolymers, such as nanoparticles, nanofibres, and hydrogels, offer enhanced drug delivery and sustained antimicrobial release within difficult-to-reach canal regions. Their high surface area and reactivity improve contact with bacteria in root canals. Common carriers include polycaprolactone (PCL), poly (lactic-co-glycolic acid) (PLGA), and chitosan. These can be loaded with antibiotics, metal ions, or even natural compounds such as curcumin or chitosan.136
NGAS are advantageous in endodontic therapy in that they offer controlled, sustained antibacterial release over days to weeks; they show Improved biofilm disruption and deeper canal penetration; and some formulations also show osteogenic, anti-inflammatory, and antioxidant effects, potentially aiding periapical healing. However, the use of NGAS is still under development with only a few published clinical studies. Nonetheless, NGAS represent a promising adjunctive therapy for AP, offering targeted antibacterial action and potential regenerative benefits.136
10. Deciding Whether to Retain or Remove Root Canal Treated Teeth (RCTT)
Deciding whether to retain RCT-treated teeth can be a difficult and stressful decision for patients to make. Numerous factors should be considered. Obvious signs and symptoms of infection such as pain, swelling, fistula formation, periapical radiolucency (PARL; seen sometimes on 2-D radiographs or 3-D CBCT), and mobility of the tooth are common criteria that a dentist will consider when advising whether to have a tooth extracted. There is a range of severity of these criteria that a dentist will discuss with the patient when making a recommendation. Sometimes, as part of an informed consent process, a dentist will mention the possibility of having a second (‘revision’) RCT performed on the tooth.
Perhaps a more challenging decision are cases wherein no pain or obvious signs of RCT problems exist. In such cases, a CBCT radiographic evaluation of a tooth and the surrounding bone is critical. An important indication of the presence of chronic apical periodontitis is a PARL as mentioned above. Depending on the size of the PARL some dentists will recommend keeping the tooth and monitoring the size of the PARL with a periodic CBCT in the absence of any symptoms.
Although not routine or accepted as the standard of care, additional tests are available. A thermogram will create a heat map of the head and neck region that reveals potential areas of inflammation. Thermography can detect inflammatory reactions at early stages of infection, leading to early diagnosis. The results, however, are general and unable to assess the health of specific teeth like a CBCT can but can be beneficial to use as a complementary technique.137,138
Blood tests that measure the levels of hs-CRP, fibrinogen, interluken-6, interluken-10, and/or CCL5 (RANTES) may offer clues about the inflammatory status within the body.139 Unfortunately, these biomarkers don’t indicate the source of the inflammation. To achieve greater diagnostic specificity, the gingival crevicular fluid (GCF) from the root canal treated tooth can be analyzed for levels of the enzyme active matrix metalloproteinase-8 (aMMP-8).140 aMMP-8 is a key mediator of tissue destruction, and its concentration in GCF can be used to assess chronic AP. Elevated aMMP-8 levels indicate the presence and severity of disease, and rapid chairside tests are becoming available in some countries for point-of-care diagnosis. However, further research is needed to establish definitive cut-off values for treatment outcomes and to clarify the enzyme’s role in differentiating between disease severities.140
11. Conclusions
In conclusion, although RCT remains an important dental intervention for managing pulpal disease, the cumulative scientific evidence indicates that RCTT frequently retain chronic apical inflammation and microbial burden that may contribute to systemic disease in susceptible individuals. CAP is common, often dentally asymptomatic, and underdiagnosed when relying on conventional 2-D radiography alone. Associations between CAP and systemic inflammatory, metabolic, cardiovascular, neurologic, and autoimmune conditions are supported by epidemiologic data, mechanistic pathways, and a rapidly expanding body of animal research showing biological plausibility. These findings underscore the necessity of moving beyond a tooth-centered model of care toward an integrated medical/dental approach that accounts for individual patient risk factors, systemic health status, and long-term biologic consequences. Informed consent, advanced diagnostics, careful case selection, and consideration of alternative or adjunctive treatment strategies are essential to optimizing both oral and overall health outcomes.
To download or print this page in a different language, choose your language from the drop down menu in the upper left of the website first. If you would like the references to print along with the position paper, please click the “References” area at the bottom of the article to expand them into view and then click the print button.
12. References
IAOMT: State of the Science on Root Canal Treated Teeth (RCTT)





