Failures in Human Subjects Protections & the Need for Federal Action
A U.S. government-funded clinical trial placed mercury-containing dental fillings into vulnerable children—many of them institutionalized orphans—while serious allegations of sexual abuse were unfolding within the very system that housed them. Despite mounting evidence of abuse and internal awareness among researchers, the study continued, raising profound questions about whether these children were ever truly protected as human subjects.
Documents obtained through Freedom of Information Act requests (as detailed by Noel Fritsch in National File) and subsequent investigative reporting by Anita Vazquez Tibau, have reignited scrutiny of the Casa Pia trial, suggesting not only a failure of oversight, but a systemic breakdown in ethical safeguards for some of the most vulnerable participants in federally funded research.
This NIH-funded study stands as a documented case of profound ethical and regulatory failure and demands immediate, independent federal review, full transparency, and corrective action to protect public health and restore trust in human subjects research.
Vulnerable Children, Systemic Abuse, and Continued Exposure
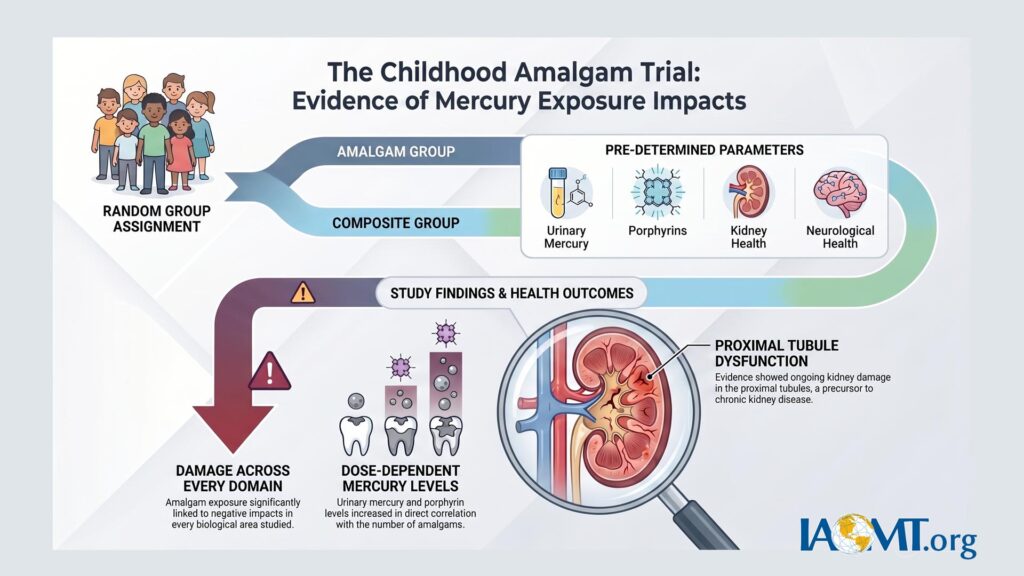
The trial enrolled approximately 507 children, many of them wards of the state residing in the Casa Pia institutions in Lisbon, Portugal. These children—aged 8–12 at baseline—received mercury-containing dental amalgam fillings during critical periods of neurological and physiological development. At the same time, they endured an environment later documented for widespread sexual and physical abuse.
Federal regulations under 45 CFR Part 46, Subpart D impose heightened protections for children, particularly wards of the state. These include requirements for independent advocates, minimization of coercion or undue influence, and strict limits on research involving greater-than-minimal risk without direct benefit (§46.406–409). Evidence suggests these safeguards were inadequately applied or enforced, raising serious questions about informed assent, oversight, and the ethical justification for continuing the study amid the known risks of mercury and reported harm.
The study proceeded without interruption, exposing vulnerable participants to mercury while they faced additional trauma. This convergence of research exposure and institutional abuse represents a stark breakdown in the principles of the Belmont Report—respect for persons, beneficence, and justice.
Scientific Evidence of Harm: Beyond the Original Conclusions
The original trial reported no statistically significant average neurobehavioral differences between amalgam and composite groups. However, subsequent independent analyses of the same dataset revealed clear signals of harm, particularly in biologically plausible subgroups and specific biological systems:
- Renal toxicity: Woods et al. (2008) identified biomarkers of kidney tubular and glomerular stress correlated with mercury exposure from amalgam.
- Heme pathway disruption: Woods et al. (2009) documented altered urinary porphyrin profiles, consistent with mercury body burden, especially in younger children.
- Dose-dependent kidney effects: Re-analyses by Geier et al. (2012–2013) found statistically significant associations between cumulative amalgam exposure and markers of proximal tubular damage, including microalbuminuria and glutathione S-transferase alterations.
- Neurobehavioral and genetic susceptibility: Woods et al. (2014) reported cognitive and behavioral deficits linked to mercury in children with specific genetic polymorphisms (e.g., CPOX4 and BDNF variants), indicating that average null results masked harm in susceptible subgroups.
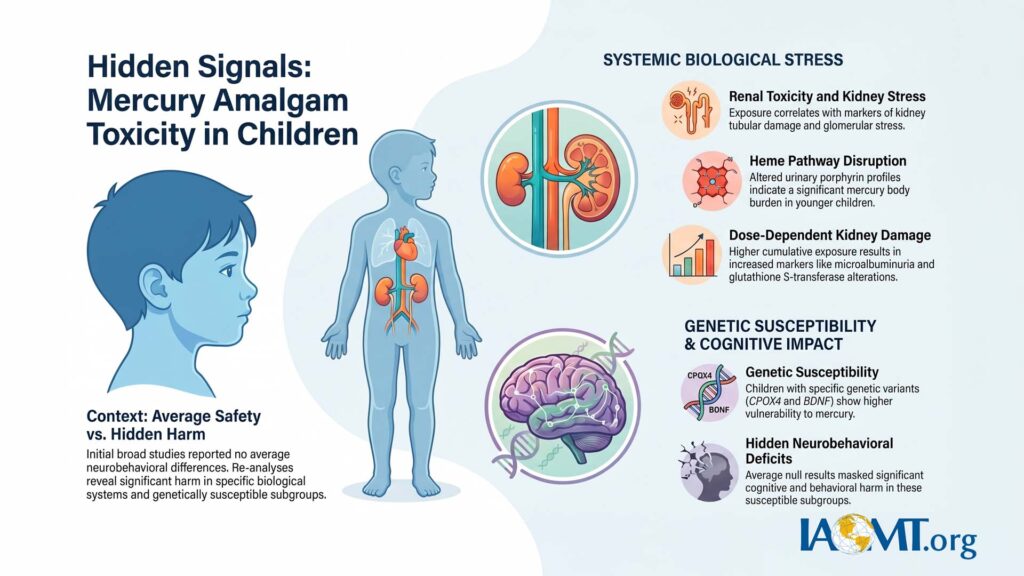
These re-analyses highlight limitations in the original study’s conclusions, including potential underemphasis of subgroup effects, dose-response relationships, and long-term implications. Clinically meaningful harm appears to have been present but not adequately highlighted in primary reporting.
Regulatory Reliance on Compromised Data
Despite these findings and the ethical red flags, U.S. agencies—including the NIH and FDA—have continued to cite the Casa Pia trial in policy statements and safety assessments supporting dental amalgam use in children. Attempts to publicize these concerns via press release were rejected by distribution services as “speculative,” even when grounded in FOIA records and peer-reviewed re-analyses.
This raises a fundamental question: Why does documented evidence of risk to vulnerable children continue to underpin public policy while calls for re-evaluation face institutional resistance?
Formal Petitions for Accountability
In response, a formal petition has been submitted to the Office for Human Research Protections (OHRP) requesting:
- An immediate for-cause compliance evaluation of the trial and associated IRBs.
- A formal determination of noncompliance with 45 CFR 46, Subpart D.
- Suspension of federal reliance on Casa Pia data in regulatory or policy decisions.
- Development and enforcement of Corrective and Preventive Action Plans (CAPAs).
- Coordinated remediation and support for surviving participants.
A parallel request for investigation has been filed with the HHS Office of Inspector General (OIG), focusing on:
- NIH and IRB oversight failures.
- FDA’s ongoing use of the data.
- Adequacy of adverse event reporting and data integrity.
- Federal response (or lack thereof) to reports of abuse during the study.
A Broader Public Health Imperative
The Casa Pia trial is not an isolated historical event. Over 180 million Americans still carry mercury amalgam fillings, creating continuous low-level exposure. Safe, large-scale removal faces significant barriers, including limited numbers of clinicians trained in rigorous mercury-safe protocols, such as the IAOMT’s Safe Mercury Amalgam Removal Technique (SMART).
The original participants—now adults—have never received systematic notification of potential long-term risks, comprehensive medical monitoring, or support for remediation. This ongoing omission violates basic ethical obligations to research subjects.
Advocates such as David Kennedy and Anita Vazquez Tibau, along with organizations like the IAOMT, continue to demand transparency, independent data re-evaluation, and justice.
Recommended Actions for Professionals and Advocates
Please share this article widely to help raise awareness of these important ethical and public health issues. Please consider emailing the Office of the Secretary at the Department of Health and Human Services (HHS) to urge federal support, notification, monitoring, and remediation for the victims of the Casa Pia trial. Contact HHS Secretary Robert F. Kennedy, Jr. at secretary@hhs.gov or call the toll-free number 1-877-696-6775.
Additionally, contact your U.S. Representative and both U.S. Senators in just a few minutes.
IAOMT members, dental professionals, researchers, and concerned citizens are urged to act:
- Demand Federal Review — Submit formal inquiries to HHS, OHRP, NIH, FDA, and congressional oversight committees regarding continued reliance on Casa Pia data.
- Support Independent Reassessment — Advocate for transparent, third-party re-analysis of the full dataset, with emphasis on subgroup effects, genetic susceptibility, and long-term outcomes.
- Advance Safe Mercury Removal Standards — Promote and expand training in evidence-based, SMART-certified protocols for amalgam removal to minimize additional exposure.
- Support Victim Remediation — Back coordinated efforts to provide:
- Notification and informed risk disclosure to participants.
- Long-term medical monitoring focused on renal, neurological, and immune systems.
- Access to safe amalgam removal where clinically appropriate.
- Consideration of evidence-based detoxification support and trauma-informed care.
- Build Public Awareness — Share verified documentation, including FOIA records, peer-reviewed re-analyses, and regulatory filings, while encouraging open scientific discourse.
Conclusion: Time for Accountability
The Casa Pia trial embodies a dangerous convergence: ethical lapses in protecting the most vulnerable, potential scientific misrepresentation, and continued regulatory reliance on flawed data. Ignoring these issues perpetuates harm to the original victims and undermines trust in federal oversight of human subjects research and dental materials safety.

About IAOMT
The International Academy of Oral Medicine and Toxicology (IAOMT) is a global network of dentists, physicians, and researchers dedicated to advancing safe, science-based dental care that promotes whole-body health.
Original Trial and Primary Publications
DeRouen TA, Martin MD, Leroux BG, et al. (2006). Neurobehavioral effects of dental amalgam in children: a randomized clinical trial. JAMA, 295(15):1784–1792. (Main neurobehavioral outcomes report; found no significant average differences.)
Bernardo M, Luis H, Martin MD, et al. (2007). Survival and reasons for failure of amalgam versus composite posterior restorations placed in a randomized clinical trial. Journal of the American Dental Association, 138(6):775–783. (Restoration longevity and failure rates.)
Biomarker and Secondary Analyses from the Original Team
Woods JS, Martin MD, Leroux BG, et al. (2008). Biomarkers of kidney integrity in children and adolescents with dental amalgam mercury exposure: findings from the Casa Pia children’s amalgam trial. Environmental Research, 108(3):393–399. (Urinary GST-alpha, GST-pi, and albumin as kidney damage markers.)
Woods JS, Martin MD, Leroux BG, et al. (2009). Urinary porphyrin excretion in children with mercury amalgam treatment: findings from the Casa Pia Children’s Dental Amalgam Trial. Journal of Toxicology and Environmental Health, Part A, 72(14):891–896. (Porphyrin profiles as indicators of mercury effects on heme synthesis.)
Lauterbach M, Martins IP, Castro-Caldas A, et al. (2008). Neurological outcomes in children with and without amalgam-related mercury exposure: seven years of longitudinal observations in a randomized trial. Journal of the American Dental Association, 139(2):138–145. (Neurological hard and soft signs.)
Additional supporting or design papers:
DeRouen TA, et al. (2002). Issues in design and analysis of a randomized clinical trial to assess the safety of dental amalgam restorations in children. Controlled Clinical Trials.
Evens CC, Martin MD, Woods JS, et al. (2001). Examination of dietary methylmercury exposure in the Casa Pia study. Journal of Toxicology and Environmental Health, Part A.
Genetic Susceptibility and Neurobehavioral Re-Analyses (Woods et al.)
Woods JS, Heyer NJ, Echeverria D, et al. (2012). Modification of neurobehavioral effects of mercury by a genetic polymorphism of coproporphyrinogen oxidase in children. Neurotoxicology and Teratology, 34(5):513–521.
Woods JS, Heyer NJ, Russo JE, et al. (2013). Modification of neurobehavioral effects of mercury by genetic polymorphisms of metallothionein in children. Neurotoxicology and Teratology, 39:36–44.
Woods JS, Heyer NJ, Russo JE, Martin MD, Farin FM (2014). Genetic polymorphisms affecting susceptibility to mercury neurotoxicity in children: summary findings from the Casa Pia Children’s Amalgam Clinical Trial. Neurotoxicology, 44:288–302. (Comprehensive summary of genetic modifiers, including CPOX4 and BDNF variants.)
Independent Re-Analyses by Geier et al. (Dose-Response Focus)
Geier DA, Carmody T, Kern JK, King PG, Geier MR (2011). A significant relationship between mercury exposure from dental amalgams and urinary porphyrins: a further assessment of the Casa Pia children’s dental amalgam trial. Biometals, 24(2):215–224.
Geier DA, Carmody T, Kern JK, King PG, Geier MR (2012). A dose-dependent relationship between mercury exposure from dental amalgams and urinary mercury levels: a further assessment of the Casa Pia Children’s Dental Amalgam Trial. Human & Experimental Toxicology, 31(1):11–17.
Geier DA, Carmody T, Kern JK, King PG, Geier MR (2013). A significant dose-dependent relationship between mercury exposure from dental amalgams and kidney integrity biomarkers: a further assessment of the Casa Pia children’s dental amalgam trial. Human & Experimental Toxicology, 32(4):434–440.
Notes:
The original trial randomized ~507 children (aged 8–12 at baseline) to amalgam or composite restorations and followed them for up to 7 years. Primary conclusions emphasized no significant average group differences in neurobehavioral outcomes, but secondary and re-analyses highlighted signals in subgroups, dose-response relationships, renal biomarkers, and genetic susceptibility.



