NEW RESEARCH LINKS DENTAL AMALGAM MERCURY FILLINGS TO ARTHRITIS
CHAMPIONSGATE, FL, June 22, 2021/PRNewswire/ — The International Academy of Oral Medicine and Toxicology (IAOMT) is raising awareness of research linking cases of arthritis to dental amalgam fillings. These silver-colored fillings are 50% mercury and are still used in the United States, often in disadvantaged children and adults. (https://desertrose.com/)
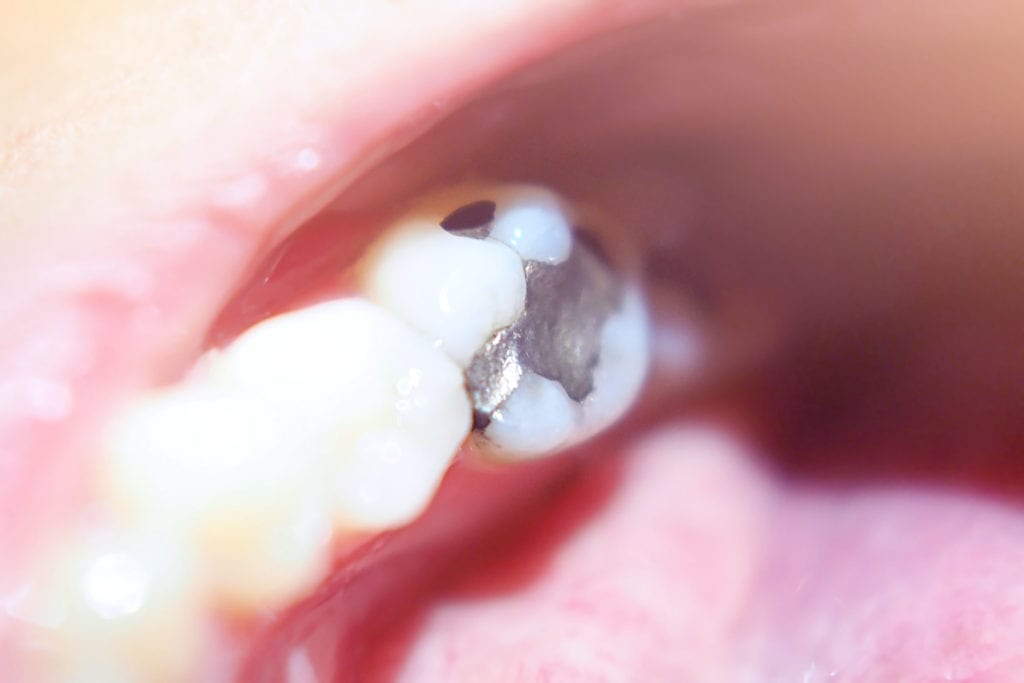
All dental amalgams are silver-colored and contain about 50% mercury. These fillings are still being used in the U.S. even though they have been associated with health risks.
In this new study, researchers David and Mark Geier report on a significant relationship between the number of dental amalgam filling surfaces and diagnoses of arthritis. They find that incidences of arthritis peak among adults with 4 to 7 dental amalgam filling surfaces.
It is important to note that the number of surfaces is not the same as the number of fillings. Each tooth has five surfaces, which means that a person with only one filling could have up to five surfaces.
The authors examined data from the 2015-2016 National Health and Nutrition Examination Survey (NHANES) including demographics, dental exams, and arthritis diagnoses. Data about the patient’s type of dental filling recently became accessible. With this information, the researchers were able to discover higher incidences of arthritis in people with silver-colored mercury amalgam fillings than those with other fillings such as tooth-colored composites.
Researcher David Geier discusses the human health risks of mercury exposure in relation to dose, disease, susceptibility, and glutathione.
In September 2020, the Food and Drug Administration (FDA) updated risks of dental amalgam fillings for susceptible groups. However, arthritis was not specifically mentioned when the FDA warned of “harmful health effects of mercury vapor released from the device.”
The groups that FDA advised to avoid getting dental amalgam fillings include pregnant women; women planning to become pregnant; nursing women and their newborns and infants; children; people with neurological disease such as multiple sclerosis, Alzheimer’s disease or Parkinson’s disease; people with impaired kidney function; and people with known heightened sensitivity (allergy) to mercury or other components of dental amalgam.
FDA is currently open for comments on how information about medical devices including dental amalgam fillings should be shared with patients and providers.
“Mercury is continually off-gassed from dental amalgam fillings,” explains David Kennedy, DDS, Past IAOMT President. “With the Geiers’ new research joining the ranks of thousands of other studies, it is abundantly clear that mercury from amalgams poses a danger to everyone, including patients, dentists, and dental employees.”
The Geiers’ study was partially funded by the IAOMT, a non-profit organization that evaluates biocompatibility of dental products, including mercury filling risks.


